 
The engine developed by Carnot has air (which is assumed to work as a perfect gas) as its working substance enclosed in a cylinder, in which a frictionless piston A moves. In a Carnot cycle, the working substance is subjected to a cyclic operation consisting of two isothermal and two adiabatic processes. He focussed his attention to the basic features of a heat engine. Miller, Computational Methods of Neutron Transport, American Nuclear Society, 1993, ISBN: 2-4.This cycle is designed by “ Carnot” who was the first scientist to analyze the problem of the efficiency of a heat engine, ignoring its mechanical details. Hetrick, Dynamics of Nuclear Reactors, American Nuclear Society, 1993, ISBN: 3-2. 
Neuhold, Introductory Nuclear Reactor Dynamics, American Nuclear Society, 1985, ISBN: 9-4. Bezella, Introductory Nuclear Reactor Statics, American Nuclear Society, Revised edition (1989), 1989, ISBN: 3-2. Department of Energy, Nuclear Physics and Reactor Theory. DOE Fundamentals Handbook, Volume 1 and 2. January 1993. Robert Reed Burn, Introduction to Nuclear Reactor Operation, 1988.Physics of Nuclear Kinetics. Addison-Wesley Pub. Nuclear and Particle Physics. Clarendon Press 1 edition, 1991, ISBN: 978-0198520467 Nuclear Reactor Engineering: Reactor Systems Engineering, Springer 4th edition, 1994, ISBN: 978-0412985317 Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. 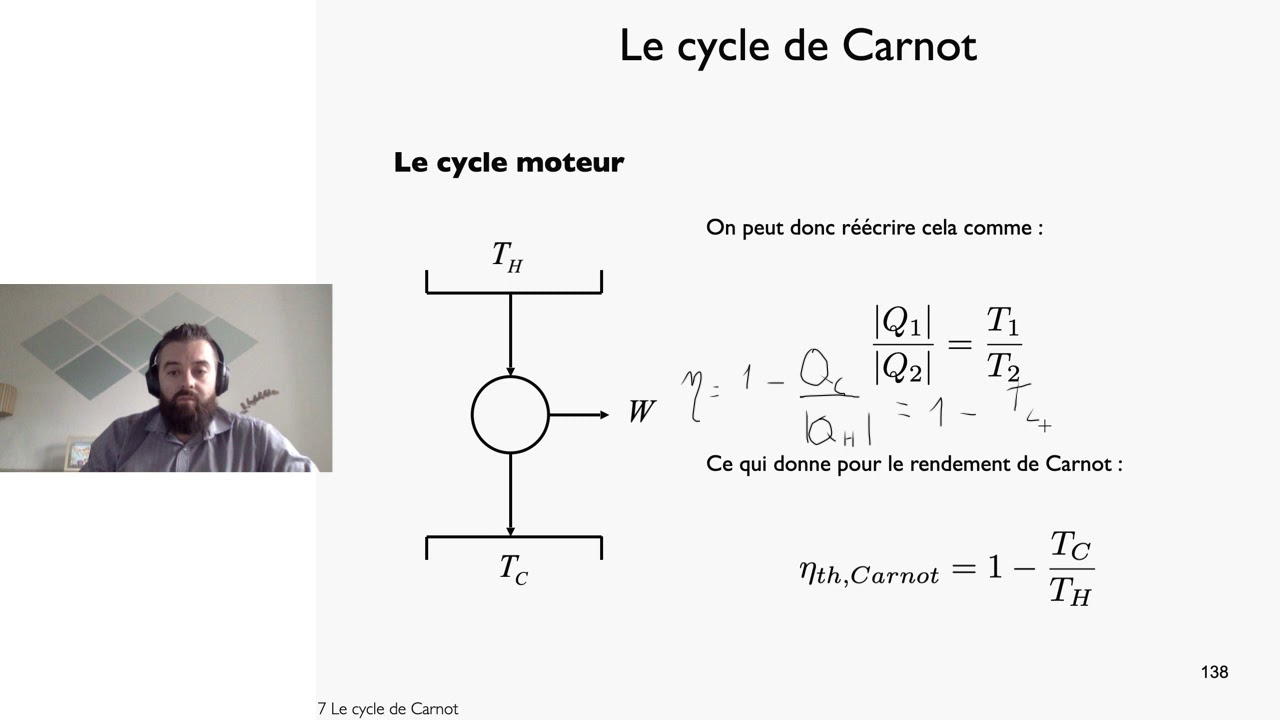
Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). The area under the Ts curve of a process is the heat transferred to the system during that process. For reversible (ideal) processes, the area under the T-s curve of a process is the heat transferred to the system during that process. It is a useful and common tool, particularly because it helps visualize the heat transfer during a process. The temperature-entropy diagram (Ts diagram), in which the thermodynamic state is specified by a point on a graph with specific entropy (s) as the horizontal axis and absolute temperature (T) as the vertical axis, is the best diagram to describe the behavior of a Carnot cycle. When plotted on a pressure-volume diagram, the isothermal processes follow the gas’s isotherm lines, adiabatic processes move between isotherms, and the area bounded by the complete cycle path represents the total work that can be done during one cycle. The Carnot cycle is often plotted on a pressure-volume diagram ( pV diagram) and a temperature-entropy diagram ( Ts diagram). The area bounded by the complete cycle path represents the total work done during one cycle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
